March 12, 2026
The last few weeks have been action-packed for HIV prevention (and for AVAC and partners), with unpacking data and discussions from CROI; injectable lenacapavir for PrEP (LEN) rolling out in a number of Eastern and Southern Africa countries; signings (and not) of “America First” Bilateral Health Agreements; and our latest quarterly STI and HIV updates. AVAC is tracking it all; see below for the key updates.
AVAC’s Year in Review — and the new Access Bridge

In case you missed it, AVAC just released its 2025 Year in Review, looking back at the organization’s impact throughout the year. The last year tested global health in unprecedented ways. AVAC showed up: helping protect hundreds of millions of dollars for global health; defending HIV programs and research; delivering trusted data, tools and analysis; and accelerating equitable access to innovations, like long-acting PrEP.
And just last week, we announced the launch of our Access Bridge partnership — a new Kenyan-based organization to work with AVAC and the field to accelerate product introduction and access.
CROI 2026
The annual Conference on Retroviruses and Opportunistic Infections (CROI) once again delivered a powerful mix of scientific progress and urgent warnings about the systems needed to turn real innovation into impact. From advances in HIV prevention, cure and treatment, new data on STI treatment and service delivery, the science pointed to expanding choice, but the discussion and debate underscored that scientific progress alone is not enough; sustainable funding, strong health systems and community leadership are essential to translate discovery into equitable access.
- Read new blogs from AVACers who were on the ground at CROI: check out Cindra Feuer’s Hot Takes & Grace Kumwenda’s My CROI Notebook.
- Read our roundups of key highlights (Days 1 & 2, Days 3 & 4).
- View recordings and resources from our community webinars with leading CROI presenters.
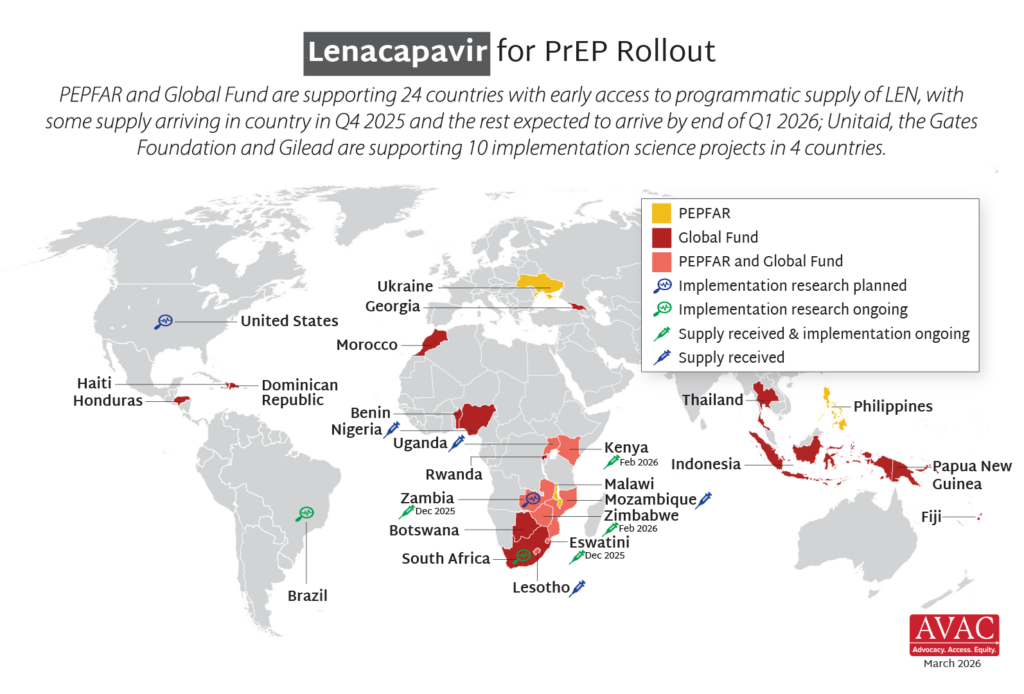
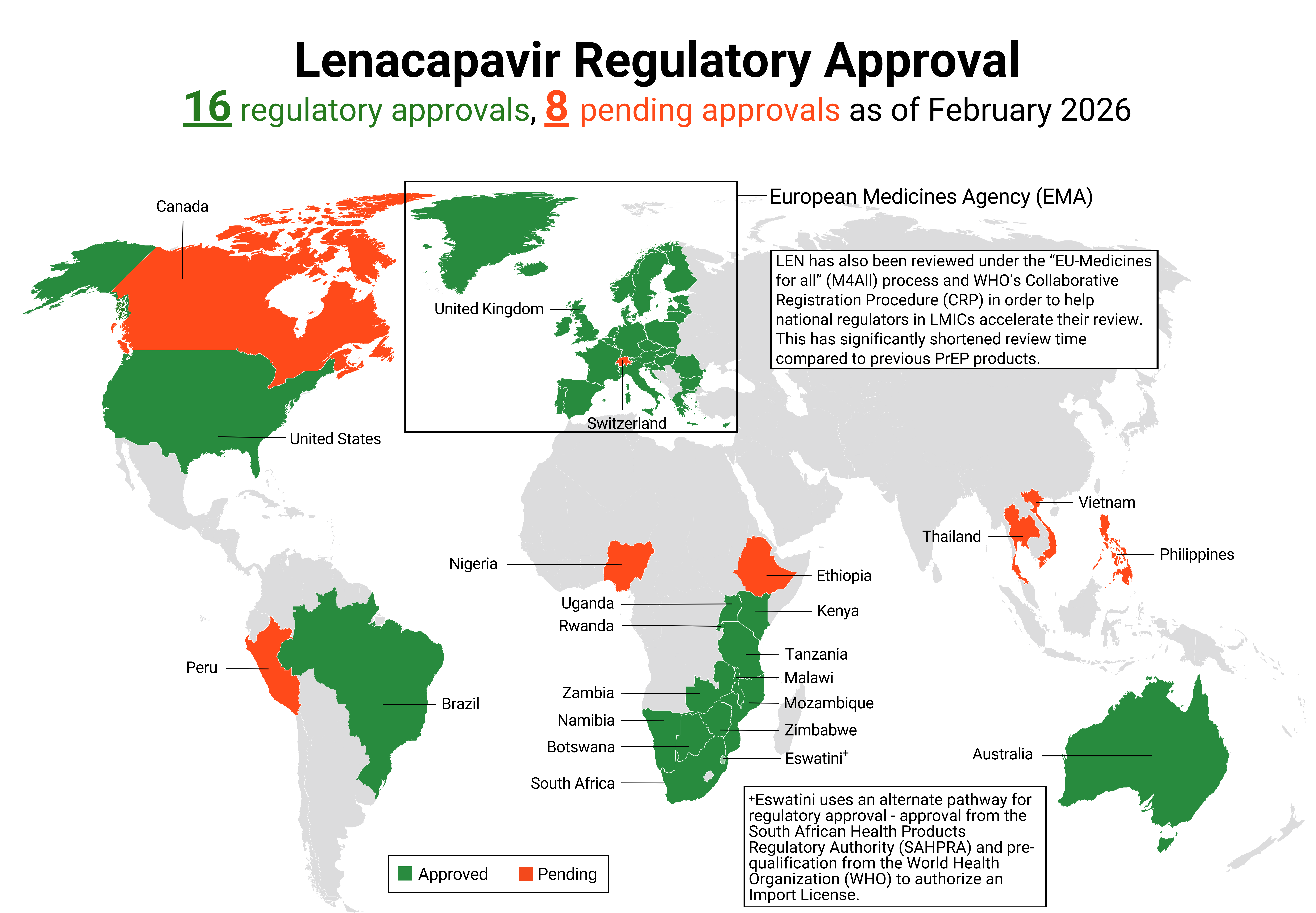
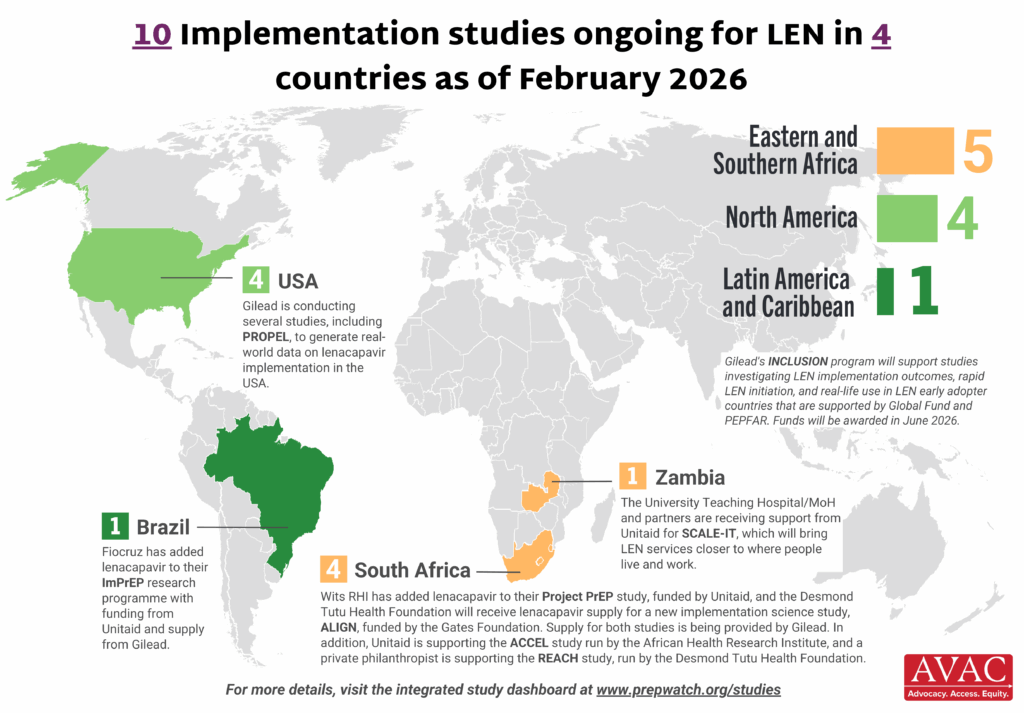
Latest Infographics: Lenacapavir Rollout Advances
Momentum continues to build around the rollout of LEN — the latest scientific breakthrough for HIV prevention. LEN delivery is ongoing in Eswatini and Zambia, it’s been launched in Kenya and Zimbabwe, and initial supply has entered Lesotho, Mozambique, Nigeria and Uganda. AVAC, Access Bridge, and the Coalition to Accelerate Access to Long-Acting PrEP continue to track the progress. Here are the latest graphics.

Tracking the “America First” Bilateral Health Agreements
AVAC Board member Stephanie Psaki examines the emerging America First bilateral health Memoranda of Understanding (MoUs) and the questions they raise around financing, transparency and the future of global health engagement in this Think Global Health piece. AVAC and partners are tracking closely developments and their implications.
Follow our Global Health Watch newsletter every Friday for the latest updates and analysis. Sign up here.
AVAC’s Quarterly Updates on HIV and STIs
AVAC’s quarterly resources bring you the latest in biomedical HIV prevention research and STIs. The latest issue of PxWire showcases the status of access to oral PrEP with the best data available since the US foreign aid freeze disrupted PEPFAR operations beginning in January 2025.
The March issue of STI Watch focuses on a growing tension in the global STI response: real scientific and programmatic progress alongside persistent underinvestment, fragile systems and geopolitics.