February 4, 2021
The annual report, HIV Prevention Research and Development Investments, which tracks funding in HIV research and development, has gone digital. The Resource Tracking for HIV Prevention R&D Working Group, a collaboration among AVAC, IAVI and UNAIDS, has launched a new website presenting 20 years of data and analysis. The website includes the latest information from the Working Group’s 16th annual analysis of HIV prevention research detailing overall investment and funding trends through 2019.
This new website offers a new tool for advocacy, with interactive analysis, and ready access to infographics. These tools can be used to advance advocacy for cutting-edge research and development and the scale-up of existing interventions needed to confront the ongoing HIV prevention epidemic, and build a stronger R&D foundation to address the COVID pandemic and whatever comes next.
Key Findings in Prevention R&D Funding
Funding is down, just a bit, but the trends are worrying.
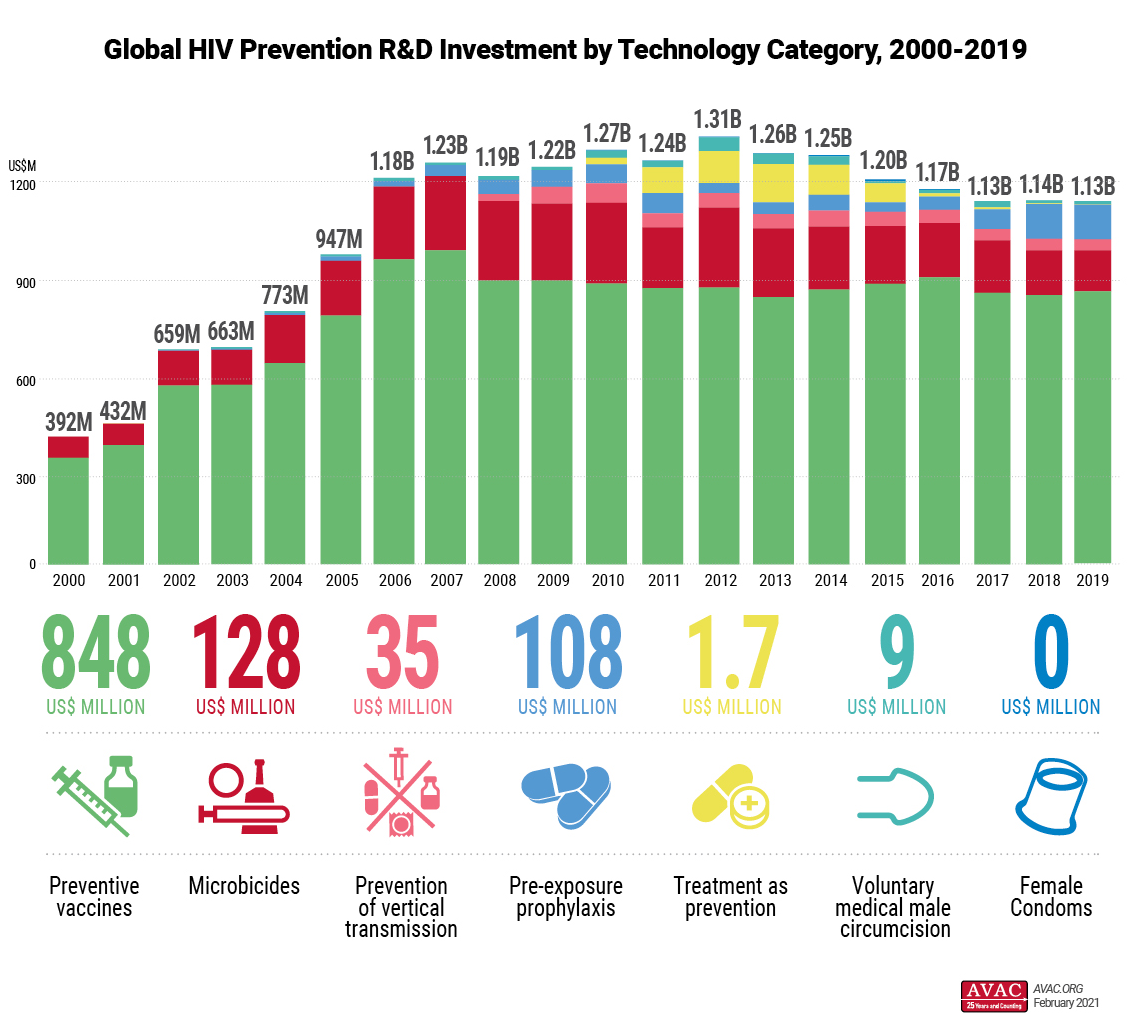
This year’s report on 2019 data found a one percent decrease in total funding, erasing the modest 1.2 increase seen in 2018 after five consecutive years of declining investment. Total 2019 funding for HIV prevention R&D was US$1.13 billion. This incremental decrease impacted the various prevention categories differently. Investment increased for preventive vaccines and female condoms but decreased for voluntary medical male circumcision, microbicides, pre-exposure prophylaxis, prevention of vertical transmission, and treatment as prevention.
Despite these differences, donor trends remained similar to 2018. Public sector (80 percent of all funding at US$902 million) and philanthropic sector (14 percent of all funding at US$158 million, a 3 percent decrease) investments remained, for the most part, unchanged from 2018. The private sector saw a 5 percent decrease in investment, falling to 6.2 percent of all funding at US$70.6 million. Actual commercial investment levels are likely higher, as some private companies do not respond to the Working Group’s request for data.
Cautionary Lessons from COVID-19 Vaccine Investment
Unprecedented investment of over US$39 billion in 2020, and collaboration and global resolve helped catapult COVID-19 vaccines from testing to human use in just over a year. As of January 2021, seven vaccines have been authorized across dozens of countries with many promising candidates in the pipeline. The process of developing a vaccine against the SARS-CoV-2 benefited greatly from early research into an HIV vaccine. Yet, rapid development of COVID-19 vaccine candidates provide both an exemplary model and a caution. Rapid R&D development is possible with investment and political commitment. At the same time, the unacceptable inequity in access to COVID-19 vaccines—with a paltry amount available on the African continent—provides a lesson for the HIV field that R&D is just the beginning of the journey towards products that actually help end epidemics. Access makes these efforts meaningful—without it research has failed, no matter the trial result.
We hope the data and new website will serve as tools for advocacy, and inform public policy that accelerates scientific progress. We thank all of the individuals who contributed data to the report and those who, most importantly, gave time and effort as trial participants.
If your organization is a funder or recipient of HIV prevention grants and we don’t know you already, please contact us at [email protected]!