During the ICASA 2023 meeting in Harare, Zimbabwe, AVAC and AfricaNPUD convened community members to draft the Harare Declaration by and for African Women Who Use Drugs.
Read the full declaration addressed to the ICASA Secretariat here.
During the ICASA 2023 meeting in Harare, Zimbabwe, AVAC and AfricaNPUD convened community members to draft the Harare Declaration by and for African Women Who Use Drugs.
Read the full declaration addressed to the ICASA Secretariat here.
This roadmap outlines a strategy for the equitable expression and delivery of HIV prevention services to key populations (KPs) globally and regionally. It introduces a critical, coordinated approach led by KPs to accelerate the implementation of existing and new HIV prevention interventions.
In the pursuit of a future without HIV and AIDS, voluntary medical male circumcision (VMMC) remains an integral component of the arsenal in HIV prevention, offering a cost-effective and scientifically proven intervention that holds immense potential in curbing the acquisition of HIV.
A two-page summary document is also available.
PxWire is AVAC’s quarterly update covering the latest in the field of biomedical HIV prevention research and development, implementation and advocacy. Each issue includes updates, emerging issues and upcoming events. Also available as a PDF.
As we look back on 2023, powerful crosscurrents confront HIV prevention and global health equity. More options than ever before could be available, but many forces threaten to undermine access to proven prevention options that exist today and the development of additional options that are still needed. Threats to PEPFAR and hate laws targeting LBGTQI+ people are just a sample.
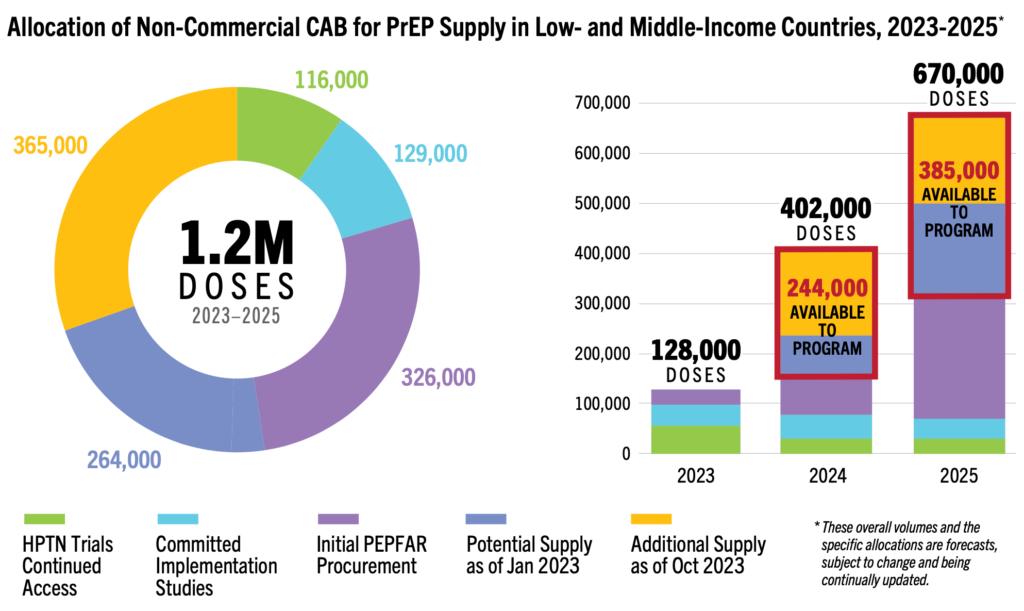
Upstream research and development is dynamic, but robust stakeholder engagement and sustainable funding must still be secured and integrated into a people-centered research agenda. These commitments are the compass that will ultimately lead to impact in the real world. The highlights below provide a snapshot of key updates in Q3 of 2023 and resources to inform advocacy on these critical questions.
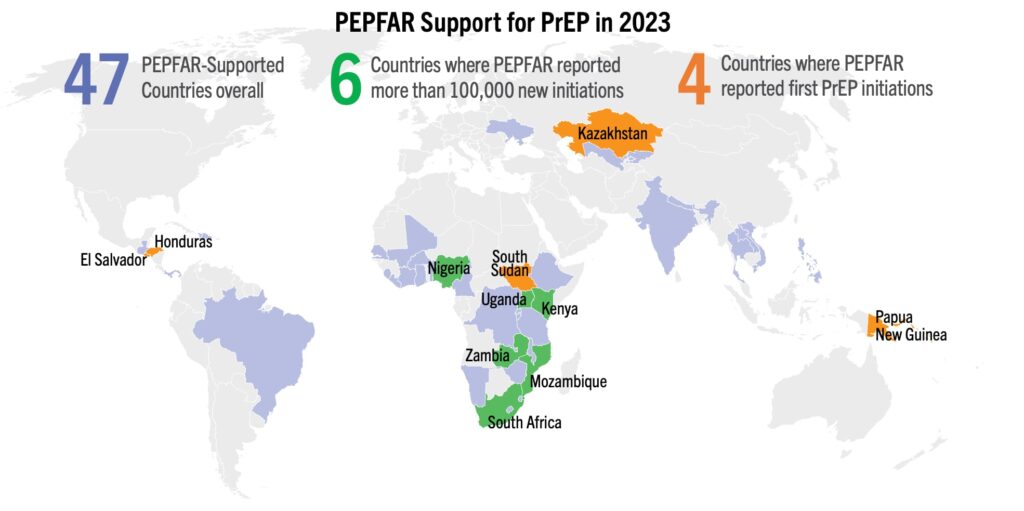
These updates from AVAC’s Global PrEP Tracker explore trends in 2023 and highlight new data as of September 2023.
PEPFAR’s role has been instrumental to accelerating global uptake of PrEP to date. This year, four countries started providing PrEP for the first time, and seven countries exceeded 70,000 new PrEP initiations, most of which are attributable to PEPFAR. This lifesaving, uniquely effective, program must see continued full funding and a 5-year reauthorization to carry on this work and help to put the world on track to control the epidemic.
Global Milestones
Country Level Milestones
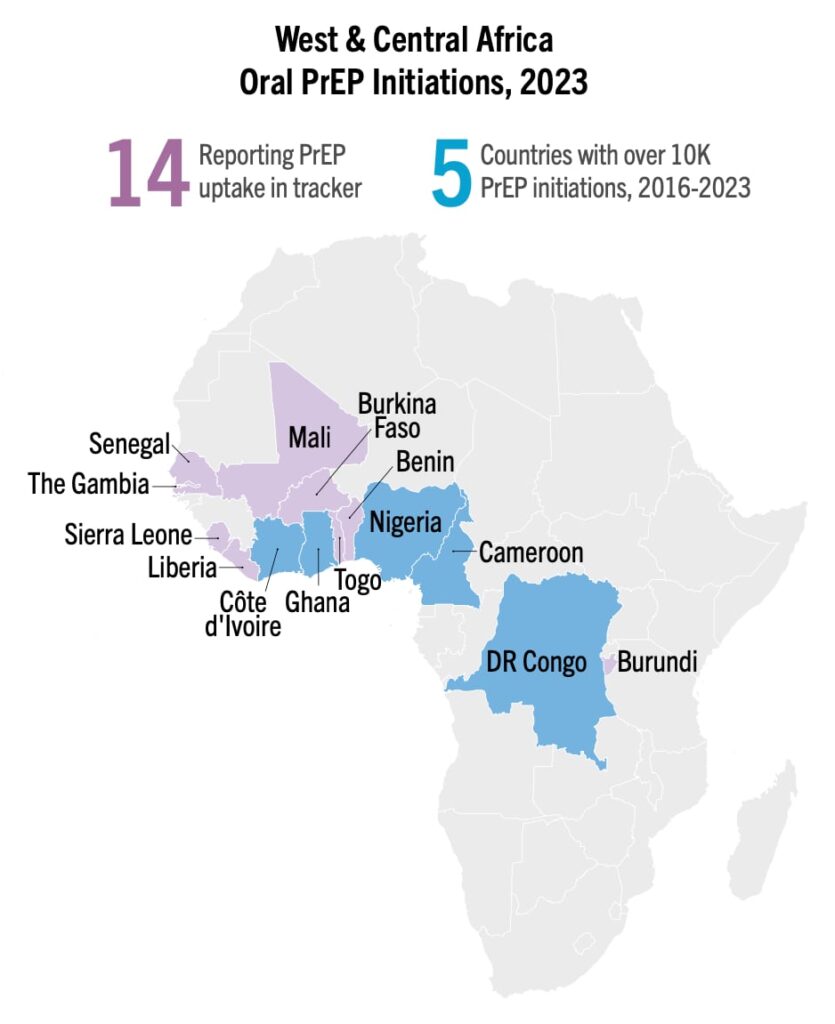
As of Q3 2023, the top five West and Central African countries for PrEP initiation have surpassed 10,000 initiations; Cameroon, Cote d’Ivoire, Democratic Republic of the Congo (DRC), Ghana and Nigeria. Nigeria has marked almost 550,000 cumulative initiations as of this quarter, ranking it second, behind South Africa, worldwide. These milestones can be credited to targeted PEPFAR investments. But despite these successes, West and Central Africa make up only 14 percent of PrEP initiations recorded in Africa, compared to 85 percent reported by East and Southern Africa. These same countries are home to two thirds of all people living with HIV in West Africa. There is vital work to be done to close the gap.
West and Central Africa can and must continue to leverage this recent growth in oral PrEP by making additional HIV prevention options available. In September, Nigeria’s regulators approved injectable cabotegravir (CAB for PrEP)—approving the first new biomedical HIV prevention option in a West and Central African country since oral PrEP. But at this time, only one other CAB for PrEP application has been submitted in the region, in Côte d’Ivoire, and none for the dapivirine vaginal ring (DVR). Only one CAB for PrEP implementation study is planned for the region.
Current HIV prevention options aren’t reaching all who need them. Access to a range of options that meet the needs of diverse people, especially those most at risk, will be essential to meeting global targets for ending the HIV epidemic.
Approval Information
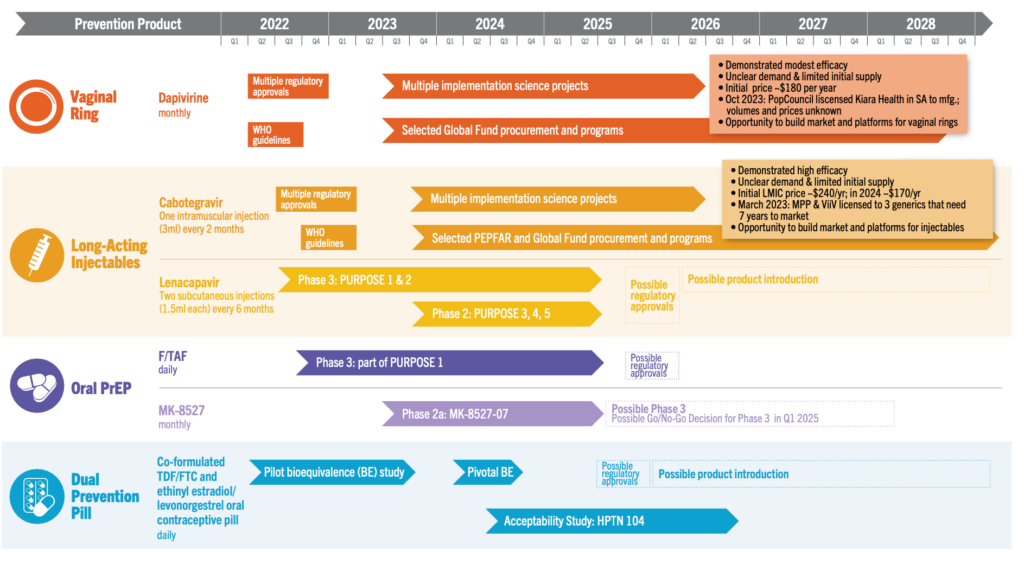
The HIV prevention pipeline has evolved: Few products are in late stage or efficacy trials, newly proven products are rolling out, and early phase clinical trials are exploring innovative strategies. R&D is focused on ARV and non-ARV based prevention products and HIV vaccines that build on new knowledge about the virus. The late-stage trials of recent years and basic science have brought deepening insights that are being applied today to a diversity of ‘upstream’ interventions.
The most effective advocacy is based on smart analysis and accurate information. AVAC develops a wide range of materials and resources to inform decision making and action. Check out these essential resources to the global conversation on HIV prevention and global health equity.
JOIN
READ
WATCH AND LISTEN
This issue brief on the vaccine research and development (R&D) process is one of a series of four briefs, which provide a roadmap for advocacy to advance the development of essential vaccines for HIV, COVID-19, tuberculosis, and other global public health threats, and approaches to ensure equitable access to these life-saving vaccines. Additional topics cover the role of mRNA technology, the need for local vaccine production, and issues around global access.
This issue brief covers the web of issues that influence access to vaccines. It is one of a series of four issue briefs, which provide a roadmap for advocacy to advance the development of essential vaccines for HIV, COVID-19, tuberculosis, and other global public health threats, and approaches to ensure equitable access to these life-saving vaccines. Additional topics cover the research and development (R&D) process, the role of mRNA technology; and the need for local vaccine production.
This issue brief on mRNA technology covers what it is, how it works, current knowledge gaps and ideas for advocacy to harness its potential. It is one of a series of four issue briefs, which provide a roadmap for advocacy to advance the development of essential vaccines for HIV, COVID-19, tuberculosis, and other global public health threats, and approaches to ensure equitable access to these life-saving vaccines. Additional topics cover the research and development (R&D) process, the need for local vaccine production, and issues around global access.
This issue brief on local vaccine production covers the current state of local production, what is needed to facilitate it, and ideas for advocacy to harness its potential. It is one of four briefs in a series providing a roadmap for advocacy to advance the development of essential vaccines for HIV, COVID-19, TB and
other global public health threats, including approaches to ensure equitable access to these life-saving vaccines once developed. Additional topics cover the research and development (R & D) process, issues around global access; and the advent of the mRNA platform for vaccines.
The FAPP AIDS Budget and Appropriations Coalition (ABAC) wrote letter urging Members of Congress to vote no on the House L-HHS Appropriations Bill (H.R. 5894), which includes $767 million in cuts and eliminations of domestic HIV programs.The letter also urges members to vote down two amendments which would eliminate funding for the Minority HIV/AIDS Fund and cut NIAID funding.
This analysis is a compilation of recommendations from civil society and community organisations on the DRAFT Negotiating Text of the WHO convention, agreement or other international instrument on pandemic prevention, preparedness, and response (WHO Pandemic Agreement).