November 5, 2024
The past few months have brought exciting developments in the field of Sexually Transmitted Infections (STIs). The World Health Organization released the global priorities for STIs, which included the need to develop low-cost, rapid, STI point-of-care tests, vaccines, and communication strategies to increase STI awareness, prevention, service engagement. But we continue to see a soaring rise in STI incidence and an underfunded infrastructure for researching new treatment, prevention and testing tools. It’s up to the global community to ensure that research and development continues to see funding for better, faster, less expensive tools to reduce the toll of STIs. Read on for resources and insights to guide your advocacy.
Conference updates
The annual conference of the International AIDS Society in July highlighted the troubling trend in the soaring rise in STI incidence. A preconference, Mobilize for Action on Sexually Transmitted Infections addressed the urgent need to confront the global spike in STI rates, particularly syphilis, gonorrhea, chlamydia, and trichomoniasis. Dr. Jeanne Marrazzo, director of NIAID, when discussing the number of global deaths from syphilis, which is treatable, said, “I think some of the more staggering statistics here, in addition to the sheer number of new infections, is the fact that we had in 2022 over 200,000 syphilis-associated deaths, which to me is practically medieval.” The meeting hosted a rigorous debate exploring the question of implementing DoxyPEP, given there’s no efficacy data for cisgender women in light of fears of creating drug resistant strains of STIs from wider use of doxycycline. Strong arguments were made on both sides, but this controversy is one of the reasons AVAC recently published an Advocate’s Guide to Doxycycline to Prevent STIs.
The STI Prevention Conference in Atlanta, Georgia convened attendees in-person for the first time in four years and discussed the rise in syphilis rates, emerging gonorrhea treatments, STI funding and policy initiatives, and doxycycline post-exposure prophylaxis (DoxyPEP). Notably, much discussion centered on NIAID’s support for a biorepository to advance diagnostic development. This biorepository would streamline access to necessary specimens to support the research and development of new diagnostics.
Save the date

New resources
Discover STIWatch.org!
STIWatch.org is an updated platform designed to enhance understanding and advocacy for STI vaccine and diagnostics research, development, and rollout. It offers comprehensive information on common STIs, a clinical trials dashboard, advocacy priorities, and a range of resources and tools to support STI prevention and treatment efforts.
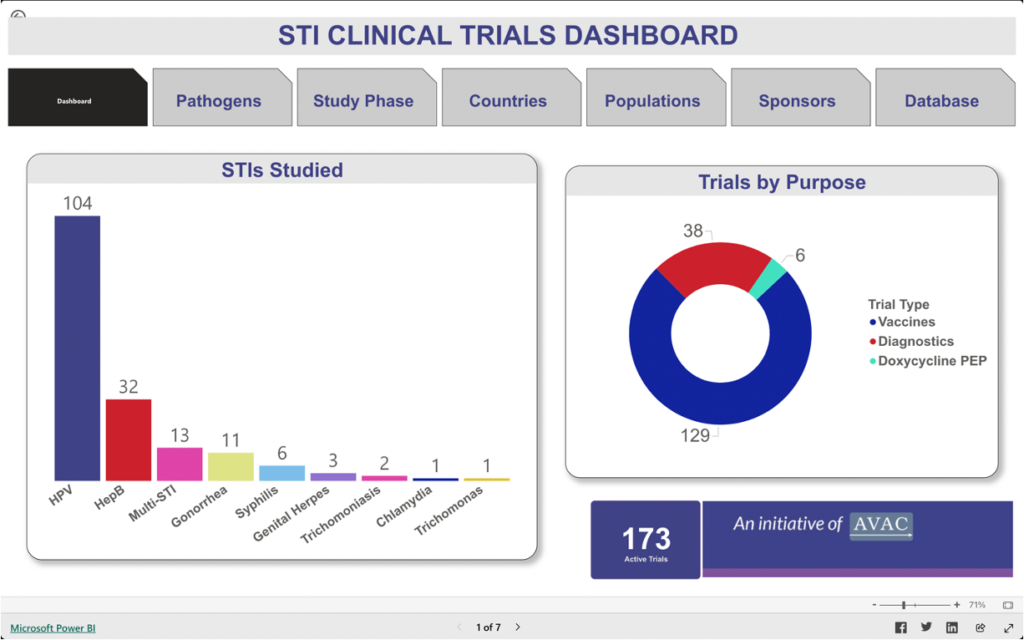
STI Clinical Trials Dashboard
This dashboard provides information about trials focused on vaccines, diagnostics, and the use of doxycycline post-exposure prophylaxis (DoxyPEP) to detect, treat, and prevent chlamydia, gonorrhea, hepatitis B, herpes simplex virus (HSV), human papillomavirus, syphilis, trichomoniasis infections, and Mycoplasma genitalium.
Webinar Summary
Regulatory Pathways to Promote Access to STI Diagnostics. This webinar co-hosted with World Health Organization (WHO) supported researchers, product developers and the global advocacy community in identifying and discussing ways to bring new STI diagnostics to market with speed, equity and scale.
What We’re Reading
- Special Issue: Sexually Transmitted Diseases. This leading journal celebrated 50 years by publishing a series of editorials that review the careers of some of the leading experts in the field. These stories are both inspiring and enlightening, highlighting the decades of efforts and growth in addressing STIs. AVAC’s own, Alison Footman, PhD, wrote about her experiences in the STI field and how instrumental mentorship has been in her career growth.
- Gonorrhea point-of-care diagnostics technology and market landscape. This landscape report provides an overview of gonorrhea point-of-care diagnostics that can be offered closer to patients and communities and limit time to results and ultimately treatment. Gonorrhea touches on multiple public health priorities including the need to reduce STI rates, growing antimicrobial resistance, and its impact on sexual and reproductive outcomes and HIV transmission.
- FDA Marketing Authorization Enables Increased Access to First Step of Syphilis Diagnosis. The US Food and Drug Administration approved the first syphilis self-test. This is a monumental step in expanding syphilis testing options as infections have increased drastically over the past five years. Self-tests can provide people with the option to learn their syphilis status and seek additional testing and treatment options from a healthcare provider.
Partner Spotlight

AVAC partners have been busy moving the needle to improve STI prevention, testing, and treatment options in their respective countries. The Latu Human Rights Foundation partnered with HEP Initiative Zambia on a symposium to foster ideas on how to better address viral hepatitis and integrate hepatitis B interventions into other government funded health programs.
To learn more about AVAC’s STI Program, visit STIWatch.org and avac.org/sti. Email [email protected] for questions or additional information. And to sign up for specific updates on STIs, click here.