Resources
AVAC’s Resource Database contains educational and advocacy materials covering a wide range of issues on biomedical prevention of HIV, STIs, COVID-19 and emerging health threats—from research to rollout.
To search for clinical trials and detailed information on products in development, visit our Prevention Research & Development Database

Results
showing 1-10 of 325
HIV Prevention Product Overview
The graphic provides an overview of PrEP products currently available and in late-stage clinical trials.

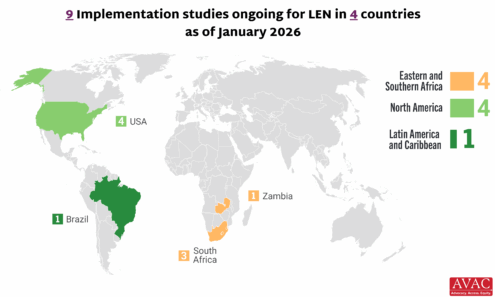
Lenacapavir Implementation Studies
Ongoing and planned implementation studies for the lenacapavir as of January 2026.
Prevention Option:
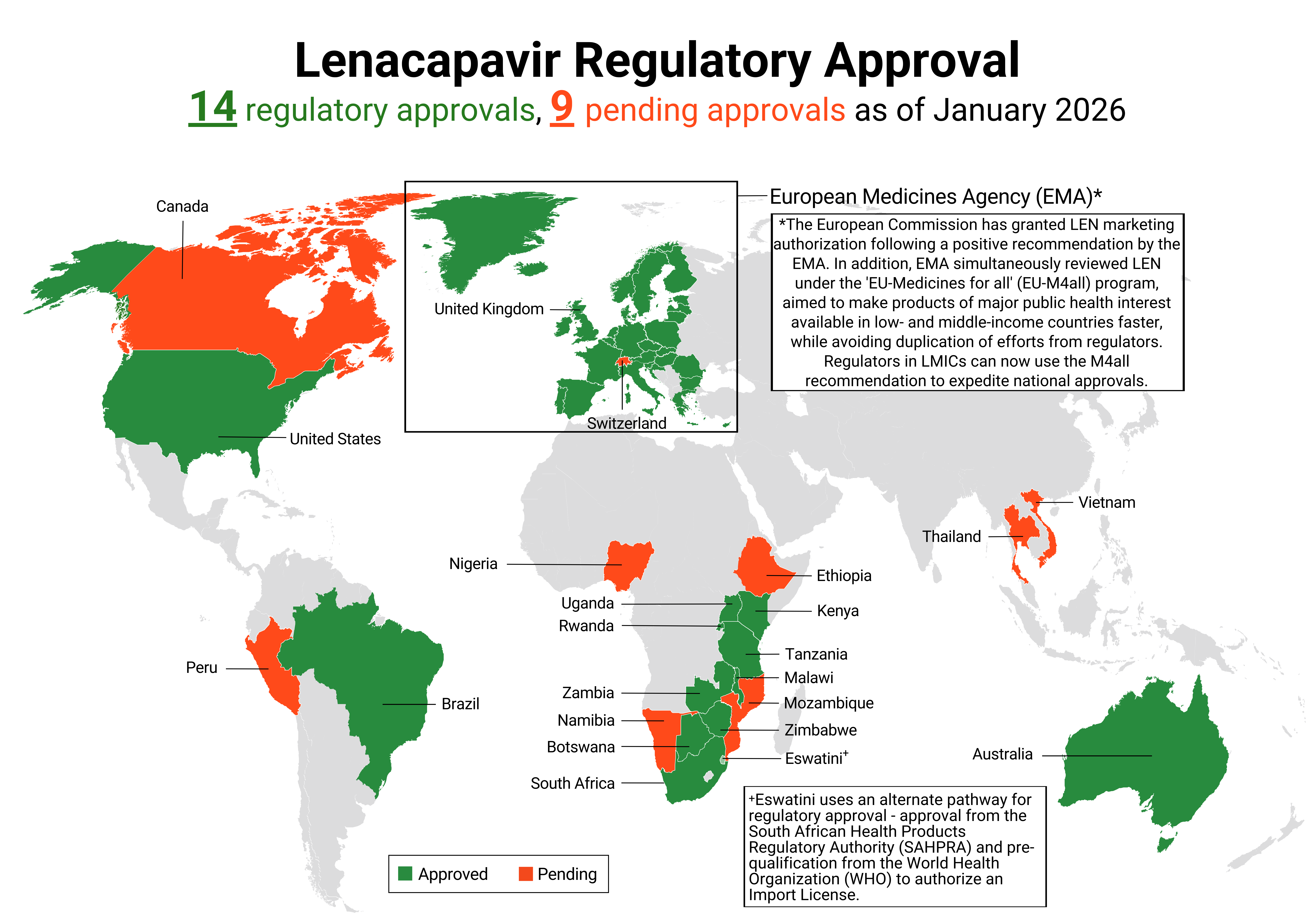
Lenacapavir Regulatory Approval
Regulatory approvals, pending decisions, and appeals as of January 2026.
Prevention Option:
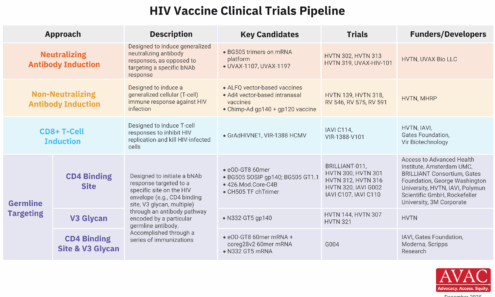
HIV Vaccine Clinical Trials Pipeline
This graphic summarizes the state of HIV vaccine research, detailing the different immunological approaches in clinical trials, the specific candidates being studied, and the collaborative networks of funders and developers working toward an effective vaccine.
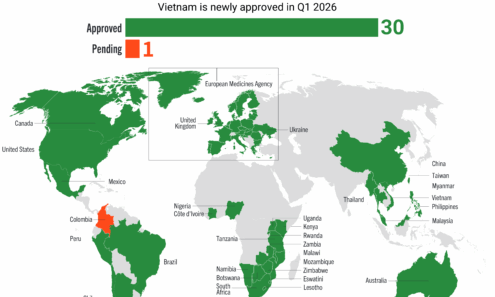
Cabotegravir Regulatory Approval
Regulatory approvals and those pending for cabotegravir as of January 2026.
Prevention Option:
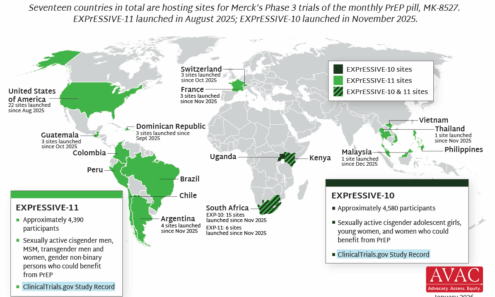
EXPrESSIVE Phase 3 Trials Countries of MK-8527
Seventeen countries are hosting sites for the Phase 3 efficacy trials of a monthly PrEP pill, MK-8527, being developed by Merck (also known as MSD outside of the US and Canada). Merck announced the launch of the Phase 3 trials at IAS 2025 in Kigali. MK-8527 was found to be safe and well-tolerated in Phase 2 clinical trials.
Prevention Option:
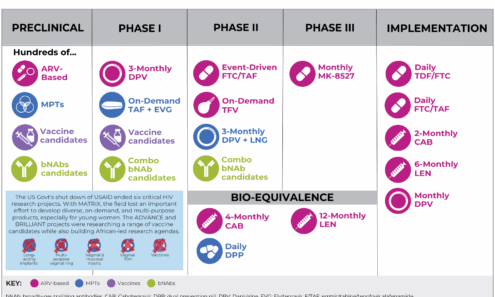
Multipurpose Prevention Technologies (MPTs) Studies Halted by USAID Funding Termination
This graphic shows a list of which studies have been halted by eliminated USAID funding.

HIV Prevention Pipeline: Products to Watch
From the People’s Research Agenda, this graphic tracks the pipeline of potential new HIV prevention options across broadly neutralizing antibodies, preventive vaccines, pre-exposure prophylaxis, and multipurpose technologies.
Prevention Option:
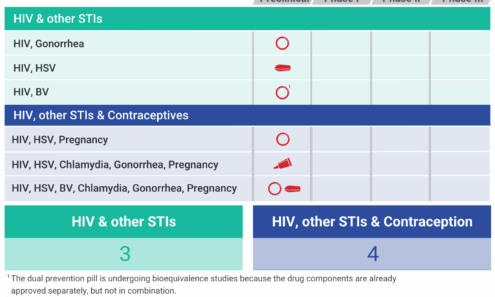
Spotlight on MPTs Addressing STIs
This graphic outlines the development journey of multipurpose technologies (MPTs) that guard against STIs, including HIV, while also preventing pregnancy. It tracks the advancement of various potential products through different trial stages, emphasizing their combined protective roles.
Prevention Option:
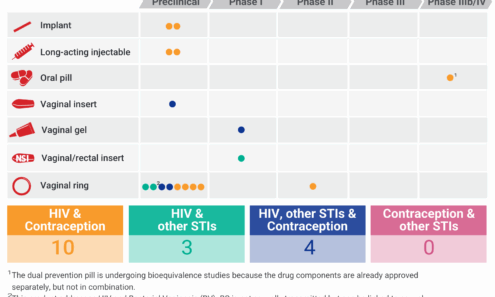
At A Glance: The MPT R&D Pipeline
This graphic shows the status of products in development.
Prevention Option:
showing 1-10 of 325