PxWire Volume 14, Issue No. 2
A Quarterly Update on HIV Prevention Research
Takeaways
- New research provides reassuring evidence that oral PrEP can reliably prevent HIV infection in cisgender women even with non-perfect adherence.
- This year, Canada, Mozambique, Tanzania, and the UK approved injectable cabotegravir, and it’s now under review in Taiwan.
- Malawi, Zambia and Zimbabwe are the second, third and fourth countries now rolling out injectable CAB outside the planned and ongoing implementation science studies.
- And much more in the issue below!
PxWire is AVAC’s quarterly update covering the latest in the field of biomedical HIV prevention research and development, implementation and advocacy. Each issue includes updates, emerging issues and upcoming events. A PDF version of this report is also available.
Progress in PrEP Uptake
New research provides reassuring evidence that oral PrEP can reliably prevent HIV infection in cisgender women even with non-perfect adherence. Prior to becoming the director of NIAID, Dr. Jeanne Marrazzo led a team in a study assessing the effectiveness of F/TDF for PrEP (brand name Truvada) among cisgender women across a wide range (11) of studies. This paper—HIV Preexposure Prophylaxis with Emtricitabine and Tenofovir Disoproxil Fumarate Among Cisgender Women, which she also presented on a recent Choice Agenda webinar—dramatically shifts the conversation away from the need for women to be “super-adherers” to oral PrEP. The data also reinforce the need to support an array of options for women.
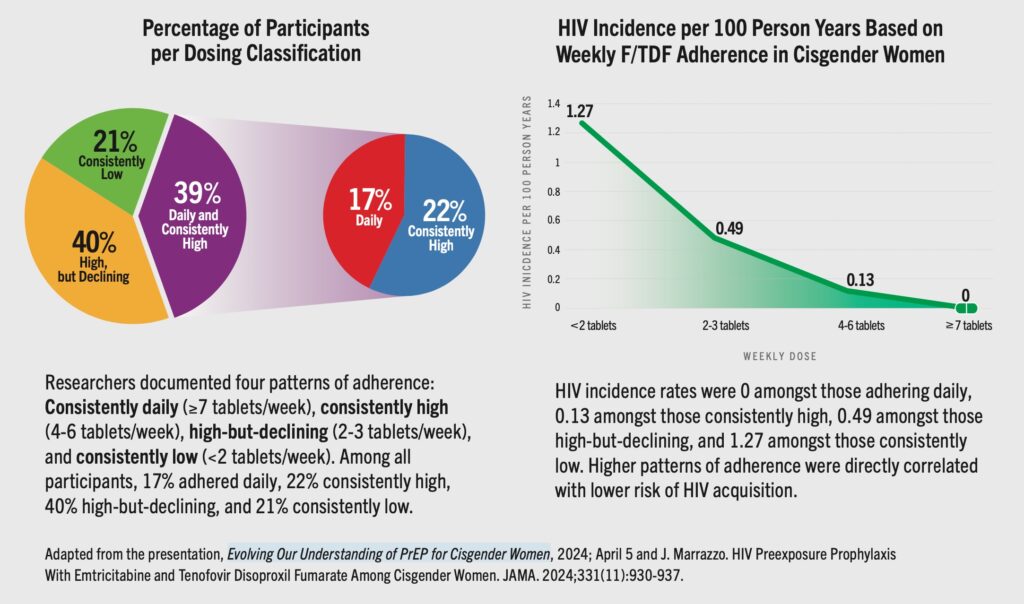
These graphics show the very high effectiveness of oral F/TDF for women with daily adherence and also for those who had consistently high adherence. Data comes from 11 demonstration projects of 2,954 cisgender women across six countries (Botswana, India, Kenya, South Africa, Uganda, USA) over eight years (2012-2020).
The data also demonstrates comparable effectiveness of F/TDF found in cisgender men who have sex with men (MSM).
Global and Country Milestones
January-March 2024
- Global cumulative PrEP initiations surpassed 6.7 million, which includes CAB initiations (12,075) and DVR initiations (1,797).
- Mexico and the United Arab Emirates approved oral F/TAF (or Descovy) in April.
For more comprehensive PrEP initiation data, see our tracker, data.prepwatch.org.
PrEParing for New Products
It has taken over a decade from positive efficacy results for daily oral TDF/ FTC to reach scale globally—and six years post-efficacy just to become available, in limited quantities, in lower-and middle-income countries (LMIC). During this time, nearly 20 million people acquired HIV. How many of those infections would have been prevented with faster, more efficient rollout of PrEP?
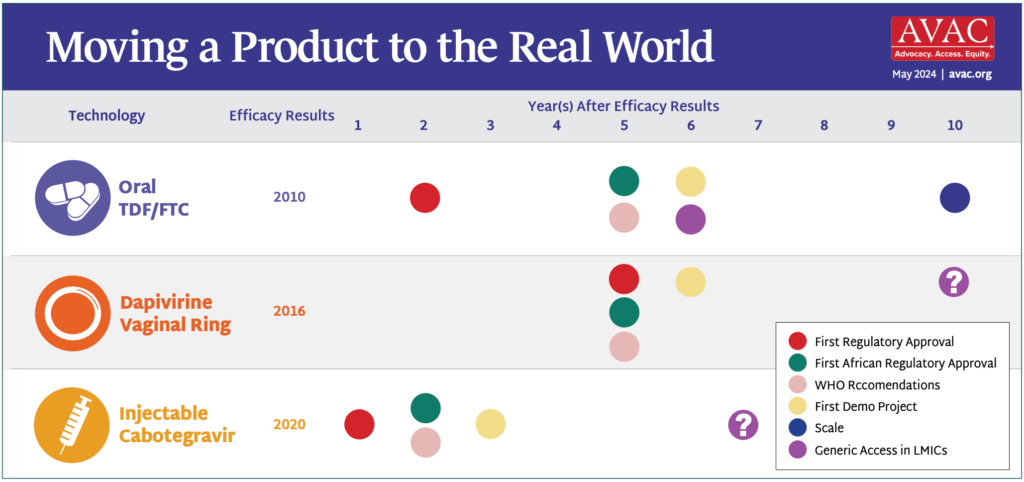
The rollout of oral PrEP demonstrates that people don’t take PrEP simply because it’s available—there needs to be a demand for it, and it needs to be accessible, acceptable and used effectively by those who need and want it. These are the lessons the field is applying to the rollout of the dapivirine vaginal ring (DVR) and injectable cabotegravir for PrEP. To reach the UNAIDS target of 10 million PrEP users by 2025, initiations of oral PrEP alone will not be enough—and the graphic at right does show that the field is beginning to apply past lessons to accelerate introduction of injectable cabotegravir.
Having multiple options for users to choose from is critical to increasing PrEP coverage, as seen from the early results of the SEARCH dynamic choice study, where coverage among participants featured a choice of products and settings for receiving oral PrEP, PEP, or injectable cabotegravir. The study showed 70% PrEP coverage in contrast with 13% coverage among those only offered oral PrEP or PEP from standard programs.
Product Updates
- So far this year, Canada, Mozambique, Tanzania, and the United Kingdom approved injectable cabotegravir, and it’s now under review in Taiwan.
- Malawi, Zambia and Zimbabwe are the second, third and fourth countries now rolling out injectable CAB outside the planned and ongoing implementation science studies. This is due to initial quantities supplied via PEPFAR.
See AVAC’s integrated study tracker for more at prepwatch.org/resources/product-introduction-country-planning-matrix.
The Latest R&D in the Prevention Pipeline
The PURPOSE efficacy trials are expected to report results later this year or in early 2025. If the six-monthly injectable lenacapavir demonstrates safety and efficacy, it will be the next long-acting injectable PrEP option to enter the market—and it is not too early to plan.
[UPDATE] June 2024: Read our blog post with the latest news, Landmark Trial in South Africa and Uganda Finds Twice Yearly HIV Prevention Injection Safe and Highly Effective

- PURPOSE 1 is evaluating the safety and efficacy of both twice-yearly injectable lenacapavir and daily oral F/TAF in cisgender women in South Africa and Uganda.
- PURPOSE 2 is assessing injectable lenacapavir among cisgender men who have sex with men, transgender men, transgender women and gender non-binary individuals who have sex with partners assigned male at birth in Argentina, Brazil, Mexico, Peru, South Africa, Thailand and the United States.
- PURPOSE 3 is studying injectable lenacapavir for PrEP among cisgender women in the United States who are disproportionately affected by HIV, with a focus on Black women and other women of color.
- PURPOSE 4 is evaluating injectable lenacapavir in adults who inject drugs in the United States.
- PURPOSE 5 will evaluate injectable lenacapavir compared to F/TDF in people who may benefit from PrEP but who are not currently taking PrEP in Europe. The study has an intentional focus on recruiting participants from groups across France and the United Kingdom that are disproportionally affected by HIV and often underrepresented in clinical trials.
Prevention Playlist
AVAC develops a wide range of materials and resources to inform decision making and action. Check out the latest:
Join
- Just What is Discovery Medicine? And What Does it Mean for HIV Vaccine Research?
- HIV, Viral Hepatitis and Sexually Transmitted Infections: Progress and gaps
- PrEP and the role of HIV self-testing
- What’s Next for the Pandemic Accord? A civil society and communities perspective?
- Global Key Populations HIV Prevention Pre-Conference
Read
- Biomedical HIV Prevention Summit and PrEP in Black America Summit: AVAC Highlights
- AVAC’s Samantha Rick delivered a statement during a high-level US HHS meeting on Thursday, April 11
- For Us by Us: PrEP in Black America—A Master Plan for HIV Prevention in Black America
- Jeanne Marrazzo and Community Leaders Amplify Their Voices for Choice
- Announcing the 2024/25 AVAC Advocacy Fellows
- AVAC Response to Uganda’s Constitutional Court Ruling on the Anti-Homosexuality Act
- STIWatch Newsletter, March 2024
- Sexually Transmitted Infections: A Review of the 2022 Vaccine and Diagnostic Research and Development Pipeline and Investments
- African Leadership and Global Health Advocacy
- Celebrating Rep. Barbara Lee and the Black Women Who Helped Shape the AIDS Movement
- HIV Cure Updates and Opportunities
- Wednesday’s CROI Roundup
- Tuesday’s CROI Roundup
- CROI Roundup—Highlights from Monday
- AVAC Condemns Ghana’s Actions to Further Criminalize LGBTQ+ People
Watch and Listen
- PxPulse: The Advocacy Chronicles with APHA’s Yvette Raphael
- Analysis & Impact of the Ugandan Constitutional Court Ruling on HIV Services
- Decolonizing Global Health: Dr. Madhukar Pai and COMPASS Africa Tell Us Why and How
- We Can’t End HIV in the United States Without Equitable PrEP Access: Strategies for success
- The Importance of HPV Vaccination Among People Living with HIV
- Discussing Early Results from the SEARCH Dynamic Choice Study
- STI Awareness Week Webinar Series
- Watching the Watcher: Intersections of surveillance and criminalization in HIV and reproductive health care
- The More We Know: Evolving our understanding of PrEP for cisgender women
- Decolonizing Global Public Health: Exploring the how, from now til as long as it takes
- Updates on Pediatric HIV Cure Research From CROI
- MOSAIC Global PrEP Learning Network—Building a Brand for PrEP: Positioning PrEP in the Hearts and Minds of Young People
- Community Breakfast Club at CROI 2024
Use
Was this content helpful?
Tell us how we can improve the content.
Was this content helpful?
Thank you for your feedback!