The Global Fund, with support from CIFF, and PEPFAR have jointly committed to reaching up to two million people with injectable lenacapavir for PrEP over three years. Supply of LEN began arriving in countries in late 2025 with service delivery planned to start in early 2026.
Source of Lenacapavir for PrEP Supply to Early Adopter Countries
CROI Cure Reflections Video Series
Produced by PAVE (Pediatric Adolescent Virus Elimination), this weekly series of four videos provides HIV cure-related takeaways from CROI 2026.
More videos forthcoming!
The Power of bNAbs: A new frontier in HIV cure research
In this video, from the Series of Jojo, viewers are introduced to broadly neutralizing antibodies (bNAbs) and how they might one day be used in finding a functional cure for HIV.
EXPrESSIVE Phase III Program Countries of MK-8527
Seventeen countries are hosting sites for the Phase III efficacy trials of a monthly PrEP pill, MK-8527, being developed by Merck (also known as MSD outside of the US and Canada). Merck announced the launch of the Phase III trials at IAS 2025 in Kigali. MK-8527 was found to be safe and well-tolerated in Phase II clinical trials.
Avac Event
Conference on Retroviruses and Opportunistic Infections (CROI 2026)
The annual Conference on Retroviruses and Opportunistic Infections (CROI) took place February 22–25, 2026, in Denver, Colorado.
Check out AVAC’s Highlights from the Meeting
- Science, Solidarity and High Stakes for the HIV Response — Day 1 & 2 at CROI
- Meaningful Progress, Expanding Choice — Day 3 & 4 at CROI
- CROI2026: Hot Takes
- My CROI 2026 Notebook: Science, Surprises, and the Big Question of Choice
AVAC Infographics
AVAC’s visuals for presentations and discussions were seen across the conference. Access them here in an easy-to-use slide format.
Community Breakfast Clubs

Each day during CROI, AVAC, EATG, Fiocruz, TAG, and other partners hosted virtual webinars called Community Breakfast Clubs featuring researchers and advocates breaking down important prevention, treatment and cure science presented at CROI and the implications for the field.
AVAC Infographics for CROI 2026
Going to CROI? AVAC’s up-to-date infographics — on the research & development pipeline, prevention options, and the impact of US funding cuts — are available here for use in conference presentations. We constantly update these graphics, so check back for updates. The latest version of the deck will be available here.
AVAC Year in Review 2025
2025 underscored the vital role that AVAC plays in the global health ecosystem, and why our work and our partnerships have never been more essential.
This report highlights AVAC’s role as a trusted voice, a translator of science and catalyst for action and advocacy. It reflects an organization ready for the future: supporting African leadership, strengthening bridges from R&D to delivery and preparing for a new chapter as we move forward into our fourth decade as an organization. Read the PDF below or view as a webpage.
An Overview of Lenacapavir for PrEP Trials
Lenacapavir Regulatory Approval
Regulatory approvals, pending decisions, and appeals as of February 2026. For product approvals, volumes, implementation, and price comparisons of long-acting PrEP, visit our dashboard on PrEPWatch.org.
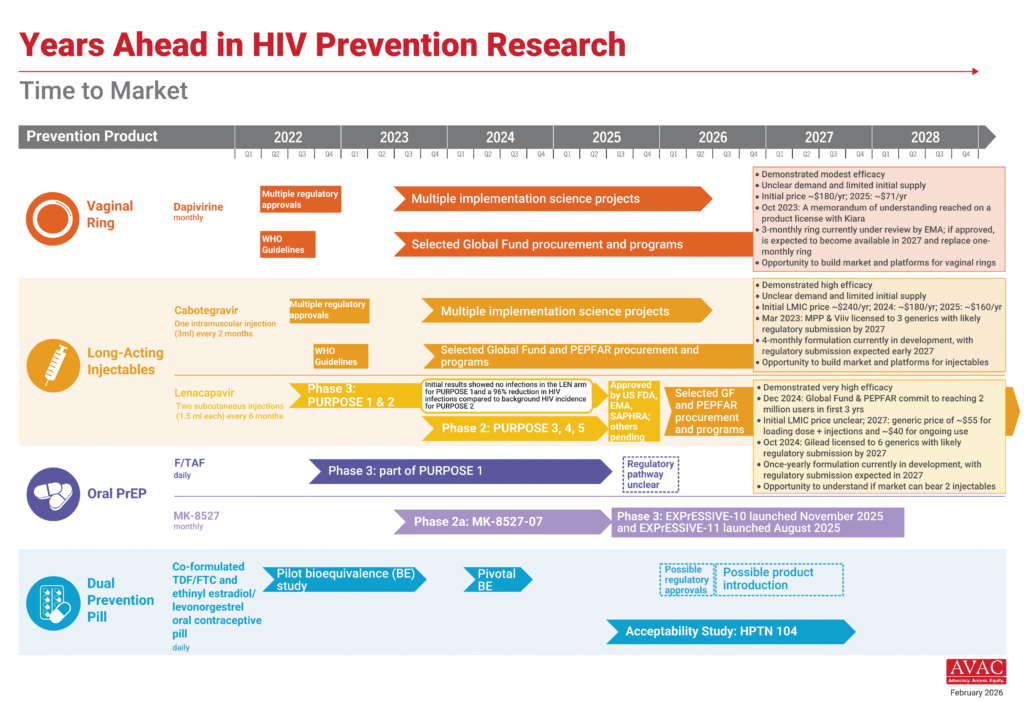
Years Ahead in HIV Prevention Research: Time to Market
This timeline shows the potential time points when the next-generation of HIV prevention options might find their way into new programs.