Resources
To search for clinical trials and detailed information on products in development, visit our Prevention Research & Development Database

Results
showing 1-10 of 999
CROI Cure Reflections Video Series
Produced by PAVE (Pediatric Adolescent Virus Elimination), this weekly series of four videos provides HIV cure-related takeaways from CROI 2026.
Prevention Option:
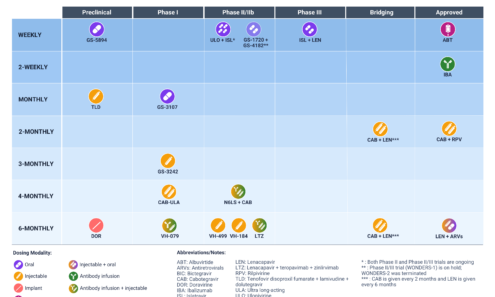
Long-Acting HIV Treatment Pipeline
AVAC has begun to track the research pipeline for long-acting HIV treatment (LA-ART). A rapidly expanding pipeline for LA-ART has the potential to introduce new regimens with dosing frequencies that range from weeks to months to, eventually, years. The benefits of LA-ART include reduced drug dosing, frequency, and number of drugs, with the potential for associated decreases in the cost of medications and the burden on health systems. New drug classes could close treatment gaps in children, adolescents, and those with high viremia, comorbidities and resistance.
Prevention Option:
PxWire Volume 16, Issue 2
This issue showcases AVAC’s analysis of the recent PEPFAR data release, assessing how uptake of prevention programs—particularly PrEP initiations—have declined over the last year. As the field grapples with the lasting impact of the US government’s cuts to PEPFAR programs, AVAC takes a closer look at what the data show and what is still missing.
Prevention Option:

AVAC Infographics for INTEREST 2026
For those presenting at INTEREST, AVAC’s latest infographics on HIV R&D and the delivery pipeline, the current landscape of prevention options are available in an easy-to-use slide format—including lenacapavir for HIV PrEP volume commitments, and the impact of US funding cuts with new data from PEPFAR.
Prevention Option:

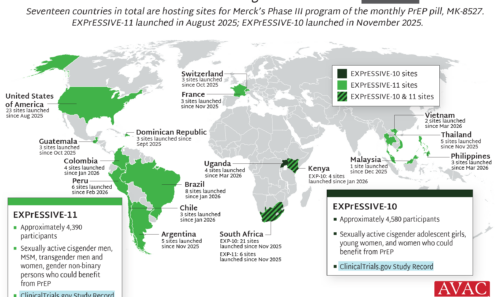
EXPrESSIVE Phase III Program Countries of MK-8527
Seventeen countries are hosting sites for the Phase 3 efficacy trials of a monthly PrEP pill, MK-8527, being developed by Merck (also known as MSD outside of the US and Canada). Merck announced the launch of the Phase 3 trials at IAS 2025 in Kigali. MK-8527 was found to be safe and well-tolerated in Phase 2 clinical trials.
Prevention Option:
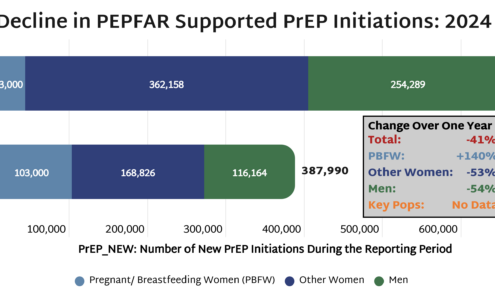
Decline in PEPFAR Supported PrEP
Official characterizations of the HIV response as “doing very good” warrant a closer look at the data. AVAC’s analysis of PEPFAR prevention figures shows PrEP initiations declined 41% in one year, with women and men each seeing drops exceeding 50%. Data on key populations remains unavailable.
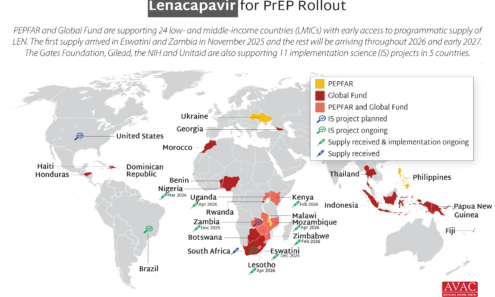
Source of Lenacapavir for PrEP Supply to Early Adopter Countries
The Global Fund, with support from CIFF, and PEPFAR have jointly committed to reaching up to two million people with injectable lenacapavir for PrEP over three years. Supply of LEN began arriving in countries in late 2025 with service delivery planned to start in early 2026.
Prevention Option:
Potential Lenacapavir Supply, 2026-28
Lenacapavir has the potential to reach millions — and the momentum is building. Using the current oral PrEP market as a baseline, this graphic estimates what demand for LEN could look like through 2028 and shows how current donor commitments stack up against that opportunity.
Prevention Option:

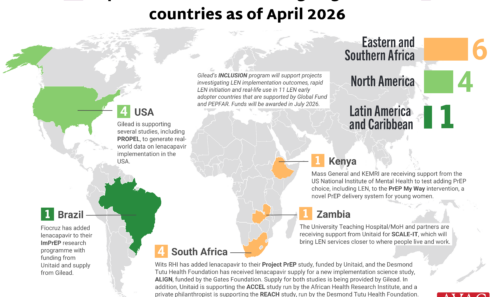
Lenacapavir Implementation Studies
Ongoing and planned implementation studies for the lenacapavir as of April 2026.
Prevention Option:
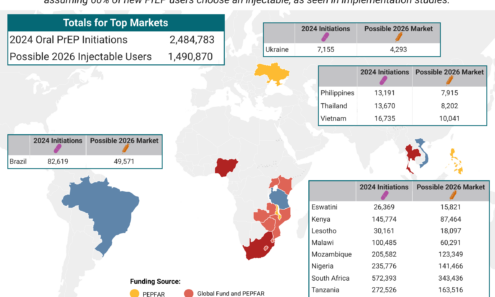
Potential Demand for LEN for PrEP
The top 16 PrEP markets, based on 2024 oral PrEP initiations and the possible 2026 injectable market, assuming 60% of new PrEP users choose an injectable, as seen in implementation studies.
Prevention Option:
showing 1-10 of 999