Resources
To search for clinical trials and detailed information on products in development, visit our Prevention Research & Development Database

Results
showing 1-10 of 1010
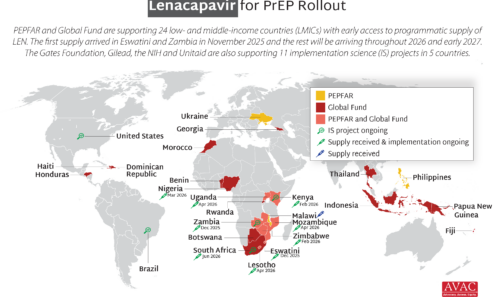
Source of Lenacapavir for PrEP Supply to Early Adopter Countries
The Global Fund, with support from CIFF, and PEPFAR have jointly committed to reaching up to two million people with injectable lenacapavir for PrEP over three years. Supply of LEN began arriving in countries in late 2025 with service delivery starting in early 2026.
Prevention Option:
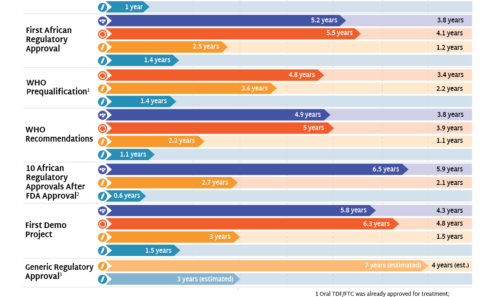
Speeding Up Access to PrEP
The timeline for hitting key milestones in product introduction is moving faster for injectable LEN than for any previous PrEP products, starting from the announcement of efficacy results in Phase III trials. LEN’s accelerated timeline compared to oral PrEP, DVR, and CAB reflects a field-wide effort to learn lessons from previous PrEP rollout and not repeat the mistakes of the past.
Prevention Option:
Advocates’ Primer: Understanding Bioequivalence and Pharmacokinetic Studies in Next-Generation HIV Prevention Products
As more next-generation PrEP options become available, HIV prevention research is shifting away from large, multi-year efficacy trials toward pharmacokinetic (PK) and bioequivalence (BE) studies — which test whether a new formulation behaves in the body like a product already proven to work. This primer explains the shift and what it means for advocates.
Prevention Option:

An Advocates’ Checklist for Lenacapavir for PrEP Introduction
The Civil Society Caucus of the Coalition to Accelerate Access to Long-Acting HIV PrEP developed a new resource that offers practical guidance to help civil society, advocates and implementers accelerate equitable access to LEN for PrEP. The checklist is designed to support faster, community-centered rollout ahead of broader generic availability in 2027.
Prevention Option:

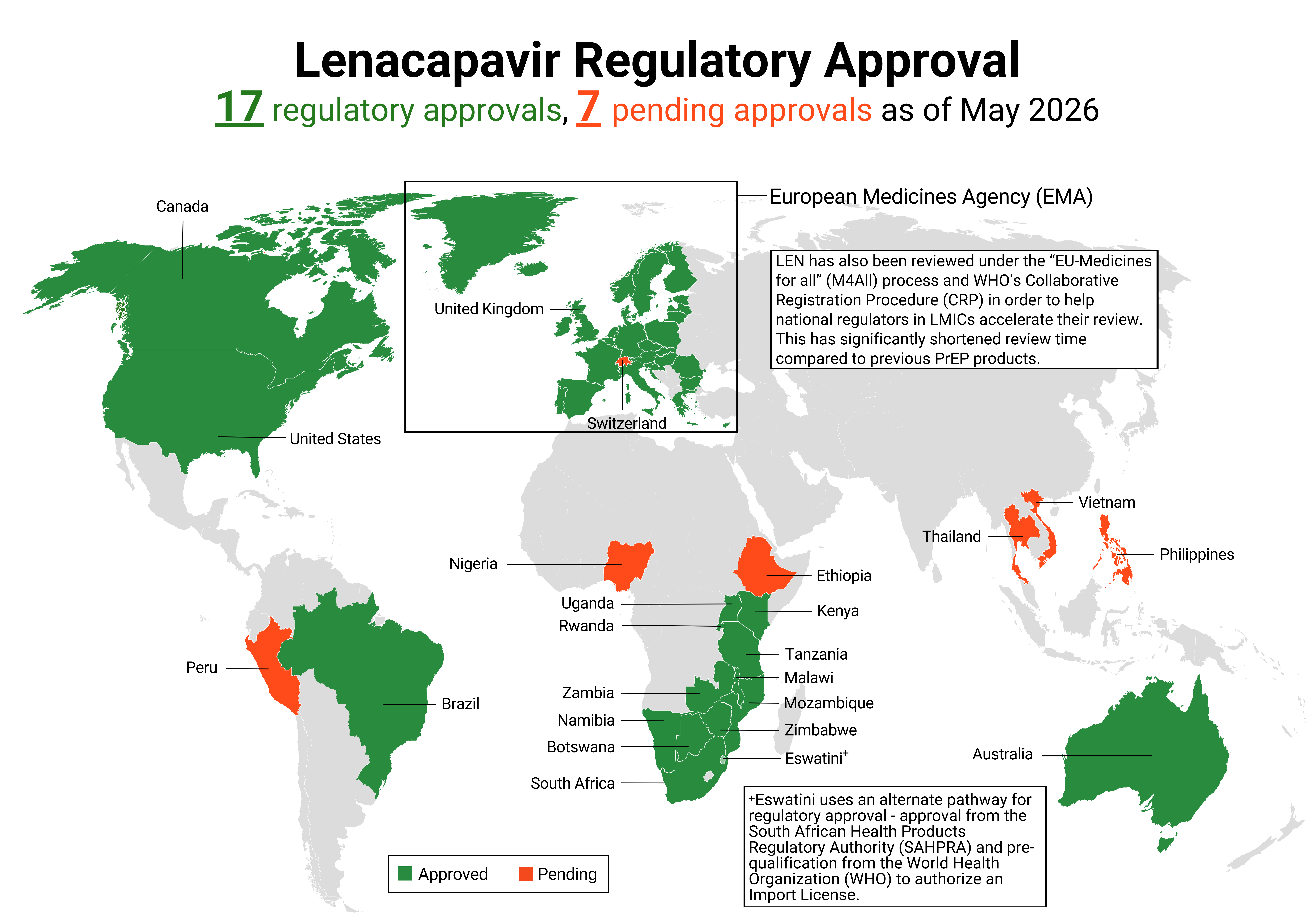
Lenacapavir Regulatory Approval
Regulatory approvals, pending decisions, and appeals as of May 2026.
Prevention Option:
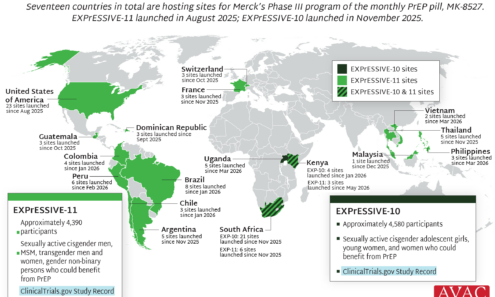
EXPrESSIVE Phase III Program Countries of MK-8527
Seventeen countries are hosting sites for the Phase 3 efficacy trials of a monthly PrEP pill, MK-8527, being developed by Merck (also known as MSD outside of the US and Canada). Merck announced the launch of the Phase 3 trials at IAS 2025 in Kigali. MK-8527 was found to be safe and well-tolerated in Phase 2 clinical trials.
Prevention Option:
PrEP Power Webinar — Global Countries
The recent significant changes to global health funding have severely impacted HIV prevention services for those who need them most, in particular key populations (KPs). Already, some countries are seeing concerning trends in data suggesting a...
Prevention Option:
PrEP Power Webinar — African Countries
The recent significant changes to global health funding have severely impacted HIV prevention services for those who need them most, in particular key populations (KPs). Already, some countries are seeing concerning trends in data suggesting a...
Prevention Option:
Analytical Treatment Interruption (ATI)
In this video, from the Series of Jojo, viewers are introduced to a methodology used to assess the efficacy of cure strategies — Analytical Treatment Interruption (ATI). Watch to learn more.
Prevention Option:

AVAC Infographics for HIV Vaccine Awareness Day
For HIV Vaccine Awareness Day, AVAC’s latest infographics — covering preventive vaccines, bNAbs, the HIV prevention research pipeline, and the impact of US funding cuts — are available as easy-to-use slides.
Prevention Option:

showing 1-10 of 1010