AVAC has begun to track the research pipeline for long-acting HIV treatment (LA-ART). A rapidly expanding pipeline for LA-ART has the potential to introduce new regimens with dosing frequencies that range from weeks to months to, eventually, years. The benefits of LA-ART include reduced drug dosing, frequency, and number of drugs, with the potential for associated decreases in the cost of medications and the burden on health systems. New drug classes could close treatment gaps in children, adolescents, and those with high viremia, comorbidities and resistance. Ongoing clinical research will hopefully further demonstrate whether there are trade-offs (treatment failures or resistance) between LA-ART and highly effective daily oral ART. In addition to the new long-acting products, simplified single-tablet daily regimens are also in development.
Long-Acting HIV Treatment Pipeline
PxWire Volume 16, Issue 2
This issue showcases AVAC’s analysis of the recent PEPFAR data release, assessing how uptake of prevention programs—particularly PrEP initiations—have declined over the last year. As the field grapples with the lasting impact of the US government’s cuts to PEPFAR programs, AVAC takes a closer look at what the data show and what is still missing.
This is especially timely, given the recent announcement from PEPFAR and the Global Fund of their additional commitment to scale-up lenacapavir (LEN) for PrEP. AVAC’s recent infographic unpacks the enduring gap between current donor commitments, LEN volume capacity and potential market size. Lastly, AVAC’s newest infographic depicts the long-acting HIV treatment R&D pipeline. Coupled with research on long-acting prevention and vaccines, long-acting treatment is an important component of an integrated approach to ending HIV as a global health threat.
Read below or download a PDF version of this issue.
Progress in PrEP Uptake
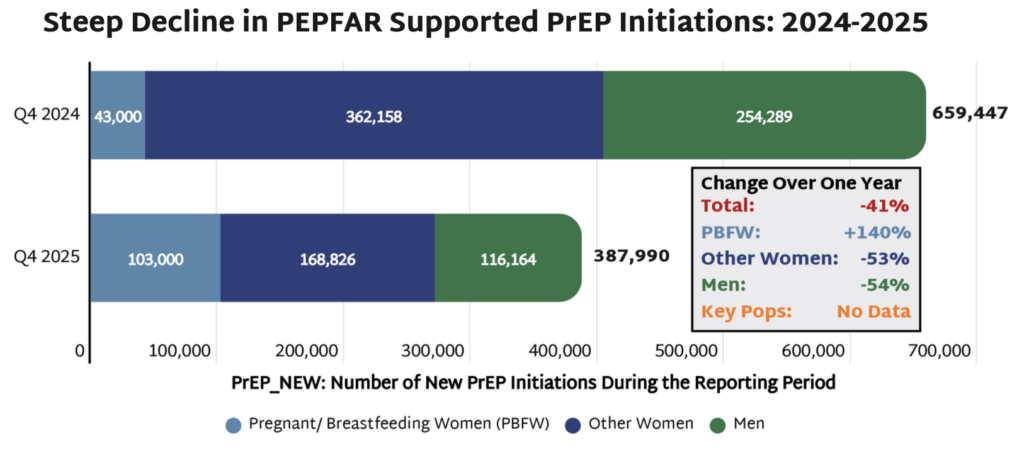
- On April 17, the US State Department released long-awaited PEPFAR data from the last quarter of Fiscal Year 2025 (FY25), representing the only data available on the status of PEPFAR programs following the US government’s drastic cuts to foreign assistance programs. Only one quarter of data, covering the fourth quarter of FY25 (July-September 2025) was released, and no data are available from the last six months of implementation.
- While PEPFAR promoted the data release as reflecting significant progress in curbing the epidemic, the data actually reveal stark declines in HIV testing, diagnosis, and treatment. HIV prevention programs were some of the hardest hit by both cuts to PEPFAR programs and the narrowing focus on prevention for pregnant and breastfeeding women (PBFW).
- In FY25, the number of individuals newly initiated on PrEP decreased by 41% as compared to the same quarter in FY24, with an overall decrease of 54% among all men and 53% among women who are not pregnant or breastfeeding. While PEPFAR touted a 140% increase in PrEP initiation among PBFW, the disaggregated data is historically inconsistent and not available in the latest data release; therefore it cannot be independently verified.
- Especially worrying was PrEP initiations among adolescent girls and young women (AGYW) aged 15-24, which fell by 39% as compared to a 26% drop among women aged 25-49. AGYW previously benefited from PEPFAR’s DREAMS programs—packages of prevention services to address the high incidence of HIV among AGYW—but the number of program recipients decreased by 86% in FY25 as many of these programs were defunded.
- Most alarmingly, the data do not include any information on HIV prevention services or PrEP initiation among key populations, including men who have sex with men, transgender people, and sex workers. This lack of data, coupled with the elimination of prevention programs for these populations, reflect an erasure of individuals at high risk for HIV that face life-threatening cuts to services.
Achieving Global 2030 Prevention Targets
The Global HIV Prevention Coalition (GPC) recently launched its HIV Prevention 2030 Global Access Framework, outlining how, by 2030, countries can ensure that 90% of people in need of HIV prevention services have access and that 90% of people living with HIV are virally suppressed. This, in combination, would lead to a 90% reduction in new HIV infections globally. “Innovations, especially lenacapavir, have added to the array of prevention choices. History will judge us harshly if we as a global community fail to meet this scientific moment,” said Mitchell Warren, GPC Co-Chair on a recent webinar. Actions must be country-led, sustainable, people-centered, and rooted in community leadership to achieve these targets.

PrEParing for New Products

- On April 14, the US State Department and the Global Fund announced an increase of their original commitment of LEN for PrEP, from two million to three million people over three years.
- Gilead has confirmed that it is not supply constrained and can supplement generic supply until it is steady, as long as they receive orders within time, based on their sixto nine-month production lead time needed. Generic supplies are expected to reach the market by the middle of 2027, but their capacity and lead times are not yet known.
- While this is an important step, it does not go far enough to have meaningful impact on rapid scale-up of LEN, and there are no guarantees that the product will reach those who need it most, including key populations and adolescent girls and young women.
- AVAC and Access Bridge see more potential to build the market by meeting market demand, leading ultimately to a reduced price, higher annual volumes and significantly increased impact.
- To achieve this, key priorities over 2026 and 2027 must be advanced:
- Global Fund and PEPFAR need to raise the floor of their commitment to reach at least four million people in two years.
- Global Fund must ensure that they have the resources to procure these volumes and meet demand, requiring resource mobilization beyond the recent GC8 replenishment.
- Global Fund and PEPFAR need to provide clear, transparent LEN access plans, with volumes and countries, ensuring a more comprehensive joint strategy in more countries, as well as volumes and prices with Gilead and generics.
- All procurers must place orders for 2027 by June 2026 with both Gilead and generics to ensure uninterrupted supply and expanded global access, whether through the Global Fund or directly with manufacturers.
- Ministries of Health must collaborate with implementation partners and civil society to ensure people-centered approaches to rollout, and to address supply and demand issues as they emerge and develop robust forecasts for 2027.
- Generic manufacturers must specify their lead-times from order to delivery to coordinate and ensure timely procurement.
- Gilead must urgently finalize license and supply agreements for middle-income countries, especially in Latin America and Asia.
The Latest in the HIV Research Pipeline
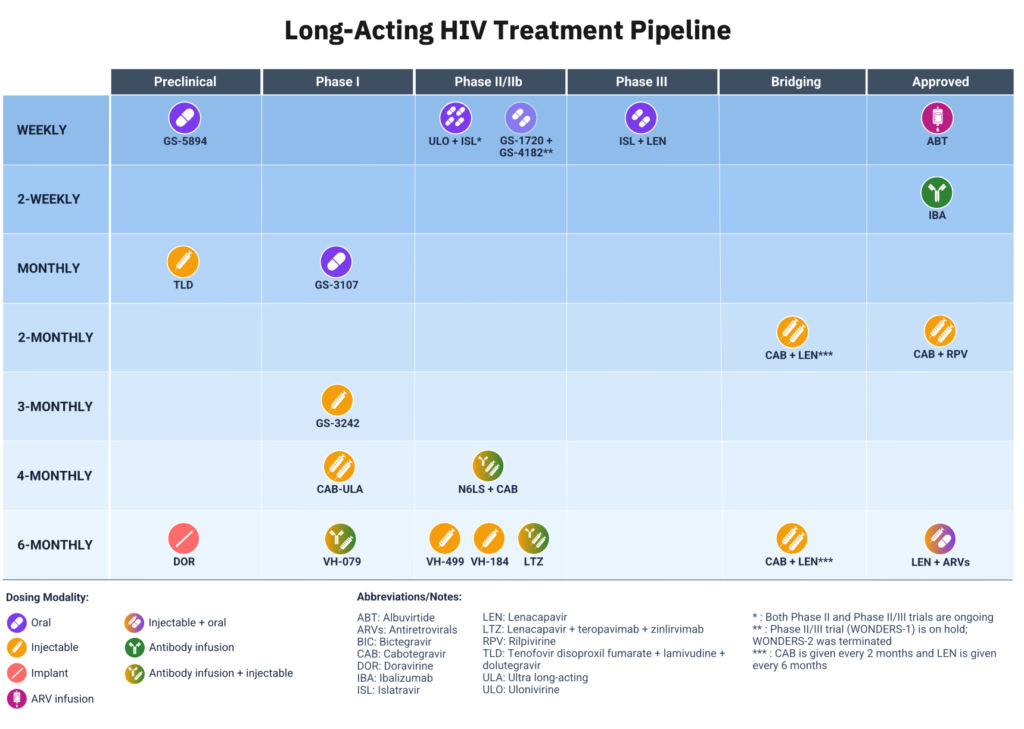
- AVAC has recently begun to track the research pipeline for long-acting HIV treatment (LA-ART). This focus aligns with AVAC’s work to advance an integrated approach to ending HIV transmission, including a comprehensive agenda for the development of novel and longer-acting products beyond PrEP, inclusive of PEP and treatment.
- A rapidly expanding pipeline for LA-ART has the potential to introduce new regimens with dosing frequencies that range from weeks to months to, eventually, years. The benefits of LA-ART include reduced drug dosing, frequency, and number of drugs, with the potential for associated decreases in the cost of medications and the burden on health systems. New drug classes could close treatment gaps in children, adolescents, and those with high viremia, comorbidities and resistance.
- CROI 2026 highlighted the potential future LA-ART landscape beyond today’s 2-month CAB + RPV injections. The range of products in development includes novel characteristics such as new ART classes and monoclonal antibodies, new ARV combinations, delivery via oral, implantable and injectable formulations, and longer regimens—currently up to six-months.
- Ongoing clinical research will hopefully further demonstrate whether there are trade-offs (treatment failures or resistance) between LA-ART and highly effective daily oral ART. In addition to the new long-acting products, simplified single-tablet daily regimens are also in development.
Building the Market for New Products from the Trial Stage
MK-8527 is currently in Phase III trials (EXPrESSIVE-10 and EXPrESSIVE-11) across 17 countries. To build a supportive environment around the concept of a monthly oral PrEP pill, AVAC supported the development of communications materials for use in the EXPrESSIVE-11 trial which can be adapted for future market entry. AVAC worked with local partners and MSM, transgender, and non-binary communities in 8 trial countries in Africa, Asia, and Latin America to co-create the materials, ensuring that focus populations contributed to the design. The result is a global campaign with a localized feel that is generating awareness on MK-8527 early.
Prevention Playlist
AVAC develops a wide range of resources to inform decision making and action. Check out the latest:
Join
- INTEREST 2026
- Launching STI Advocacy Hubs: National Agendas from Kenya, South Africa & Zimbabwe
- Sign up for Global Health Watch: AVAC’s weekly STIWatch Quarterly Newsletter – Q1 Newsletter: newsletter to keep advocates informed, prepared
Use
- Overview of Key LEN Dose, Volumes, Timelines and Prices
- STI Advocacy Agendas
- HIV Prevention 2030 Global Access Framework
Watch/Listen
- The Choice Agenda Webinar: The Injectors of Tomorrow are Here Today
- Putting communities first in HIV Research with Yvette Raphael
- Dapivirine Ring: Giving Women Hope and Choice in HIV Prevention countries in Africa, Asia, and Latin America to co-create the materials, ensuring that focus populations contributed to
Read
CROI Cure Reflections Video Series
Produced by PAVE (Pediatric Adolescent Virus Elimination), this weekly series of four videos provides HIV cure-related takeaways from CROI 2026.
More videos forthcoming!
AVAC Infographics for INTEREST 2026
For those presenting at INTEREST (as well as anyone interested in tracking the field), AVAC’s latest infographics on HIV R&D and the delivery pipeline, the current landscape of prevention options are available in an easy-to-use slide format—including lenacapavir for HIV PrEP (LEN) volume commitments, and the impact of US funding cuts with new data from PEPFAR. We hope these are useful for presentations and discussions at the conference and beyond.
We constantly update these graphics, so check back for updates. The latest version of the deck will be available here.
EXPrESSIVE Phase III Program Countries of MK-8527
Seventeen countries are hosting sites for the Phase III efficacy trials of a monthly PrEP pill, MK-8527, being developed by Merck (also known as MSD outside of the US and Canada). Merck announced the launch of the Phase III trials at IAS 2025 in Kigali. MK-8527 was found to be safe and well-tolerated in Phase II clinical trials.
Decline in PEPFAR Supported PrEP
Official characterizations of the HIV response as “doing very good” warrant a closer look at the data. AVAC’s analysis of PEPFAR prevention figures shows PrEP initiations declined 41% in one year, with women and men each seeing drops exceeding 50%. Data on key populations remains unavailable.
Source of Lenacapavir for PrEP Supply to Early Adopter Countries
The Global Fund, with support from CIFF, and PEPFAR have jointly committed to reaching up to two million people with injectable lenacapavir for PrEP over three years. Supply of LEN began arriving in countries in late 2025 with service delivery planned to start in early 2026.
Potential Lenacapavir Supply, 2026-28
Lenacapavir has the potential to reach millions — and the momentum is building. Using the current oral PrEP market as a baseline, this graphic estimates what demand for LEN could look like through 2028 and shows how current donor commitments stack up against that opportunity.
Lenacapavir Implementation Studies
Ongoing and planned implementation studies for the lenacapavir as of April 2026. For product approvals, volumes, implementation, and price comparisons of long-acting PrEP, visit our dashboard on PrEPWatch.org.
Potential Demand for LEN for PrEP
The top 16 PrEP markets, based on 2024 oral PrEP initiations and the possible 2026 injectable market, assuming 60% of new PrEP users choose an injectable, as seen in implementation studies.



